|
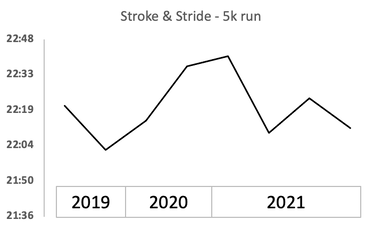
The Stroke & Stride is a local race series for those needing a mid-week reservoir swim (half mile or mile) and 5k run. There are 9 races throughout the summer and most folks use them as time trials or practice for their upcoming triathlons (swim, bike, run). I started doing these races back in 2019, during my first summer in Boulder. This upcoming week will be my 9th race, perhaps I will be able to beat that 22 minute barrier.
Summer evenings in Boulder can be quite hot, 90-95F. The temps make the swim extremely pleasant while the run can be challenging. By the time I hit the turn around point, I've mentally gathered all of the athlete goddesses I've known over my many years of training and racing. They run beside me and in front of me. I tie my imaginary knot onto them, let my love for my ladies fill my soul, and as a team, we push hard to the finish. No one ever mentioned that one of the best parts of growing up is observing the development of your friends' professional careers. Team leaders become nurses and teachers, pain-cave survivors become naturopaths and forest service rangers, dance instructors, musicians, and a lawyer. For 10 minutes each week, when I am exhausted and begging for water, these people come back to me. Suffering together, or is it pleasure? I'm still not sure. At the finish, I reach for my watermelon slices, my ghost friends are gone - back to their lives, spread out all across the U.S. and beyond. I have a half ironman (1.3 mile swim, 56 mile bike, 13 mile run) on September 11th! Woot woot.
0 Comments
We finished our Colorado Trail journey in Leadville at the end of segment 10 (Mount Massive Trailhead). We had gathered a badass girl gang to hike with, which felt fantastic. But, at the end of the day, we decided to call it quits. For future CO Trail adventures, we plan to day-run shorter segments and weekend-backpack the longer segments. We are really looking forward to the southern sections - unfamiliar terrain will entice us next year.
Day 1 on the trail was full of anticipation. We started around 8 am and hiked the dirt road through Waterton Canyon with 8 days of food weighing on our backs but spirits very high. We reached our camp at about 2 pm and set up camp just as a rain storm hit. We rode out the storm in our tent and only later realized we could see Denver skyscrapers in the distance.
The days since the first are quite a blur. I feel lucky to have quickly shifted into trail brain - a calm mindless meditation that filters out all the mental nonsense and focuses instead only on the intense flower colors and the balance between human and tree breaths. We’ve had few wildlife encounters so far - 2 moose, 3 deer, 1 fox, 2 bull snakes. And tonight we are quietly nestled into a hostel in the gorgeous Breckenridge area. A 2500’ climb in 3 miles waits for us tomorrow. And it sure will feel like a challenge with our food sacks freshly stocked with 4 days of food. I am so lucky to be headed off on the Colorado Trail tomorrow with Meg Kent from Laramie and all the other hikers we will meet out there. We have been planning and prepping for a while. And the anticipation is at its peak! Today we mailed out our four boxes of food off toward Silverton and Monarch. Meg's pack is starting at ~35 pounds and mine is close to 50! Below you can find 8 days worth of my trail food for the first portion of our journey. Highlights include a variety of pudding flavors and Thai Peanut Slaw from Farm to Summit out of Durango. I am really looking forward to those meals. We are planning for all weather conditions and are excited for what's to come. You can find updates here on this blog once per week (I hope).
Happy trails to you... Is it Christmas?!
No. It's just some of the waste produced by me during my PhD. These are just the tubes and tips from EPS and DNA extractions over the course of a year. Would be great to develop zero-waste laboratory solutions... Now that DNA has been extracted, I can move on to quantifying how much DNA is present in each sample and then how much of a particular gene is present in each sample. To quantify the total DNA concentration, we use a Qubit. The Qubit assay includes a dye that binds to DNA and then the Qubit detector measures the amount of dye bound to DNA. Once I know the total DNA concentration for each sample, I have dilute each to the same level before doing quantitative Polymerase Chain Reaction (qPCR). For qPCR, we choose a gene of interest, often the 16S rRNA gene, and we amplify that gene from every single organism that has it. We can use a dye (similar to the Qubit) to learn how much the gene amplified and backtrack how many genes were there to start. This is helpful because you can learn much more about which organisms are in the sample and how abundant they are without having to do the more intensive sequencing process. In my case, I want to know how many cyanobacteria are in the soils. It is really easy to visually estimate the abundance of mosses and lichens at the soil surface, but it is much harder to accurately estimate the number of cyanobacteria. That is why qPCR comes in handy!
I am doing all of these steps for multiple different projects. In some cases, I won't do anything else with the DNA. The qPCR is the end-point of the analysis. In other cases, I will use the qPCR data to determine which samples to sequence (which samples are worthwhile spending the additional time and money to learn about the microbial communities in depth). The past two weeks have been laboratory heavy. Which I love! My goal is to quantify the amount of cyanobacterial genes in the biocrust samples I have in order to develop a smart plan for sample analysis. If I know the number of cyanobacteria present at each step of a restoration process, then I can more easily say which step causes a significant decline. We can then zero-in on that step and focus our money and time on that part of the process. In order to quantify cyanobacterial genes, I am using qPCR with cyanobacteria-specific primers. In order to do that, I need to extract DNA from the soil, quantify the total DNA in each sample, and then do the qPCR. In the photos above, I show how the samples are homogenized (with mortar and pestle) and then extracted for DNA through many steps. I'm always shocked at how many tubes are required for these extractions (photo on the right). Here, I am extracting only 4 samples (in replicates of three) and it requires 6 tubes per sample and just as many pipette tips. Because of this large plastic waste, I am extremely careful to not mess up the extraction and I double check that every sample is worth the effort. I'm looking forward to a month from now when I can share this data with collaborators.
This week, I am measuring the texture of the various soils I have sampled from many different projects. Soil texture is important for dryland studies because finer grained soils can hold more water longer. We also find that biocrust recovery occurs more quickly on fine grained soils. We take ~13 g of each soil and remove carbonates and organics. We then use a 53 um sieve to separate sand from the finer grains. The sand can be seen above in aluminum tins. We then separate the silt from the clay by letting the silts fall out of solution (~5 hours). We can then pour off the water and clay, keeping the silts in the bottom of the beaker. We can then calculate the percent sand-silt-clay and classify the soil textures based on the standard system. By doing this analysis at the plot level, we have a more detailed understanding of what is happening in our plots rather than relying on large scale soil texture maps. These 16 samples took me 6 days of lab work, so I hope it is worth it!
Research Questions: How do altered soil moisture and nutrients influence the microbial community composition of a degraded dryland soil? Does the altered microbial community composition affect native plant germination and growth? Project Description: Claire Karban designed this field experiment and we set it up Fall 2020. Claire is testing various approaches to dryland restoration including soil pitting (shown in the photo above), biochar, and seed coating. I am curious whether these treatments have an indirect effect on native plant establishment through altering microbial communities. In particular, we expect that the pits will improve soil moisture status and will collect nutrients via dust collection. This may alter the microbial community structure. We would also like to know whether that altered microbial community structure contributes to seeded plant establishment. In the spring, I will be collecting soils from the pits, from control plots, and from healthy nearby soils. I will be extracting an intact microbial community (using methods from Corinne Walsh in Noah Fierer's Lab) from each soil type, applying them to the native seeds we used in the field experiment, and measuring plant growth in growth chambers and in the greenhouse. I will follow-up with marker gene sequencing for both bacteria and fungi. I'll link my findings to the field site, where Claire and Sallie will be measuring plant success (plant densities and biomass) for each treatment as well as fungal infection rates. Our overall goal is to improve dryland restoration strategies in ways that can be used at a large-scale.
Research Question: What are the microbial community outcomes of using an assisted migration strategy for biological soil crust restoration with inocula from three different North American deserts and cultivation at a single site on the Colorado Plateau? Project Description: Working with the USGS in Moab, I will be assessing the microbial community composition of inocula that was created as a mix of various intact biocrust communities from the Colorado Plateau, Mojave, and Sonoran Deserts. This inoculum was cultivated in the Colorado Plateau, so we might expect the inoculum to become more similar, genetically, to other local communities. Alternatively, we might predict that the more tolerant hot-desert microbes from the Sonoran Desert could out-perform those of the Colorado Plateau. If that is the case, then there may be promise for using this assisted migration technique in biocrust restoration projects.
|
AuthorSierra is a graduate student in the Barger Lab at CU Boulder studying microbial ecology for dryland restoration. Archives
August 2023
Categories |














 RSS Feed
RSS Feed
